
In today’s article, we’ll look at the management review process.
Periodic reviews of your management system can help assess its effectiveness helping you to ensure that it’s still aligned and appropriate to your business as well as helping you meet ISO9001:2015 requirements.
In this article, we’ll cover
- What’s a management review
- What is the management review process
- How to conduct a management review meeting
- How to document the Management review Process?
- An Example management review agenda
- The benefits of management review
- Some issues to look out for
Please note that we’ve written this article with relevance to ISO 9001. If you’re looking at other standards, while there are some similarities, please validate your best course of action from that standard.
Why review your Quality Management System
The basic principles behind a Quality Management System are to:
- Define policies and plans
- Implement the system (your QMS)
- Validate performance through auditing and measuring
- Introduce changes reacting to analysis and ensure continued alignment to business goals and strategy
The management review process sits with points three and four, helping to validate performance and to ensure continuous alignment to your business strategy
What is a management review meeting?
The management review process under ISO 9001 (Clause 9.3) requires an organization’s Quality Management System to undergo periodic reviews by its management/leadership.
This is to ensure it is
a) Fit for purpose by reviewing performance
b) Effective by determining the need for change
c) Suitable and appropriate for the organization
With regards it’s effectiveness, change could be on several fronts, for example:
a) Quality Policy
b) Goals & Objectives
c) Any other areas that may require change
In terms of ISO 9001, specific clauses exist for management review purposes, under ISO 9001:2015 it’s clause 9.3 Management Review.
Why have a Management review
The purpose of the management review is that it should drive continuous improvement of both the organization’s QMS and its associated manuals.
Performance is directly related to your QMS’s effectiveness; therefore, appropriate review cycles should drive an increase in both efficiency, and it’s ability to be effective. This is especially true where it builds on data obtained by your management system such as:
- Audit findings
- Corrective Actions
- Preventative Actions
As well as reviewing alignment to your business and performance, management review, when used effectively, can be a fantastic enabler helping to communicate
- What the system is
- How it works
- Recent Updates/changes that have been made
Who should attend the management review?
There are many ways of approaching both the frequency of reviews and required attendance. Whilst many think of management reviews taking place once a year, this is not so, and you may approach it in various ways. You’ll usually find a mix of the following:
- Senior Management with executive responsibility
- Line management,
- Principle process owners,
- Auditors
It is unlikely, for most organizations, that all senior managers will be available or present. The key, however, is to have a specified team, ensuring there is sufficient senior Management present to ensure that decisions can be made.
Management Review example agenda
As we’ve described, the key purpose behind management review is that it should drive continuous improvement of your QMS.
As such, it’s essential to carefully consider the agenda for your review.
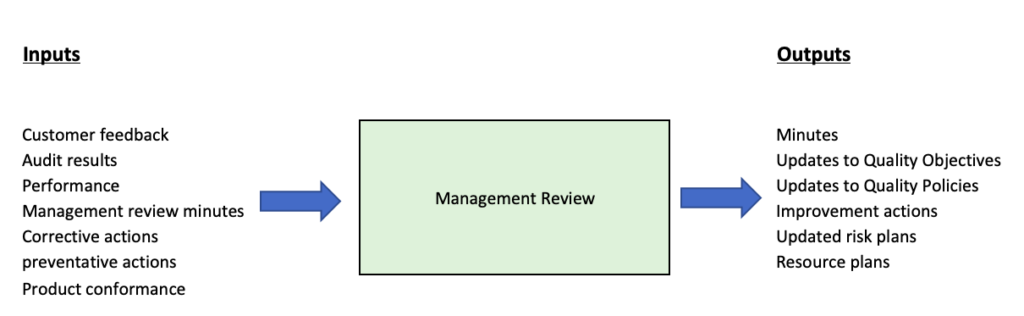
When you’re defining what should be reviewed, there’s a whole load of things that can be included. ISO9001, however, is fairly explicit in terms of expectations, and you should consider the following in terms of the basics:
- Review of minutes/actions from the previous management review meeting
- Quality Policy & Objectives
- The review should include internal & External issues that affect the business.
- The performance of the Quality Management System, including:
- Customer requirements & feedback
- Performance (i.e have targets been met)
- Alignment of quality plans & policies with company strategy
- Potential changes to QMS
- Review of Regulatory requirements
- New
- Existing
- Potential future changes
- Audits and Audit results
- Audit Schedule
- Matters arising from Audits
- Conformance of products & Services
- Preventative actions & planning
- Improvement plans
- Suppliers and External Service providers
- Risk & Opportunities
- Review of existing mitigation actions and discussion of new ones required
- Resource planning
How to conduct a management review meeting
Firstly, before you begin, it’s a good idea to document your management review process. This can make life simpler, and it should help describe:
- Your schedule & agenda
- Inputs & Outputs
- Key responsibilities
Ensure that you send a calling notice with an agenda to attendees. Certain members of the team may wish to attend for certain aspects (which may apply to them) and not for others.
Ensure that all involved with the process fully understand requirements from Clause 9.3. Remember, management review is there to serve your business first, but be aware that management review counts towards your certification.
Tips for effective management reviews
Remember, you’re reviewing your organization’s management system, which may turn some people off. Management reviews can be inspiring; however, if approached in the right way, Here are some ideas on how you can do that:
1. Be clear about the process and objectives
Management review might sound daunting and ‘exclusive’ to many unfamiliar with the process; however, it can provide a fantastic opportunity to garner staff involvement. You can help by:
- Keeping things simple
- Being clear about the task and how it will be done
- Ensuring your management system has clear management review objectives.
- Involving appropriate levels of Management to ensure that the system is practical, effective and providing value
- Seeking input from across the organization.
2. Ensure you have a structure to your process
- Have a clear agenda and don’t go off track
- Take minutes (an ISO requirement)
- Use minutes from previous reviews to review and address previous actions
3. Prepare
- Drive better engagement by:
- Provide relevant information to attendees in advance
- Being clear about your expectation of attendees
- Avoid surprises
4. Avoid falling into lecture mode
- Keep things interactive
- Seek engagement
- Look to Prompt debate
- Be upfront with participants ahead of the meeting and request involvement as them to prepare something to be discussed as part of the agenda.
How often should we have management review meetings?
There are two common myths associated with management review
- There must be one annual review.
- All requirements must be covered in one meeting.
In actual fact, ISO state that the frequency of reviews should be defined by the QMS.
As a minimum, aim to have a review at least once a year, however, best practice would indicate that it’s best to avoid one large meeting. You can have a series of reviews that add value rather than just undertaking for the sake of accreditation purposes.
Ideally, your reviews should be appropriate to your organization’s complexity and the levels of risks that you face.
Certain aspects lend themselves to be reviewed monthly (i.e., performance, customer feedback) where others, i.e., policies, may be addressed with less frequency (six-monthly). This is especially true where waiting months for decisions to change could have a detrimental impact on the business.
You could also choose to have varying levels of reviews, with each review covering several subjects. Each session could and utilizing various inputs.
For example, your supply chain team might review performance at a lower level on a monthly basis, this may also be reviewed by senior Management on a quarterly basis.
What’s important is that your organization must define the frequency within the QMS.
Another key aspect is that where a corrective/preventative action must be taken, your management review process must show that appropriate follow up action was taken and any changes to the QMS effectively implemented. When tracking improvement actions, an annual meeting isn’t necessarily constructive.
You could adopt a quarterly frequency, for example, or you may choose to combine with other business activities (it might naturally align itself to strategic planning reviews, for example).
A flexible approach to your frequency helps drive engagement within the organization by:
- Keeping the length of each management review meeting focused on key targets,
- Helps cover all required inputs
- Supports the analysis of appropriate data allowing for trends to be visible and reviewed.
Document your management review meetings
As a reminder, it’s mandatory to document your management reviews; this should include actions and observations.
ISO stipulates that all management reviews must be documented.
In reality, this means that you need to capture:
- Observations,
- Outcomes to decisions and actions,
- Further actions agreed
- Where corrective actions have been agreed, evidence should be present to demonstrate that the action was performed.
In terms of audit to ISO 9001, Auditors will require evidence from the relevant clauses (9.3 under ISO 9001:2015) under the standard.
Practically, an auditor will ask for evidence the meetings are taking place by way of documented outputs. These could include:
Typical outputs might include:
- Management review meeting minutes
- Updates to quality objectives
- Updates to Quality policies
- Documented Improvement actions for:
- Processes
- QMS
- Product
- Resource
- Business plans
- Budgets
- Updated risk management plans
You’ll need to retain your meeting minutes (as evidence for your QMS).
Note that the management review process does not replace your existing corrective action process, and any items requiring non-conformances or corrective actions should be addressed in the usual way.
Measuring management review
Now that we’ve described the process, the next question we’ll address is exactly how you find out if your management review process is delivering value.
Whilst some businesses may focus on the need for management review for the sake of ISO accreditation, it goes without saying that the primary reason for undertaking management review is that it actually helps your business!
So how do we assess that the management review process is delivering value to us? The answer, of course, is that we measure it.
We can measure the process by evaluating the effectiveness of the output of the process.
Decisions made on things like resources, risk, budgets, policies etc. can all be measured to ensure they are driving improvement and helping the business achieve its targets.
Benefits of an effective Management review process
Over time, changes can occur in business; this can be in aspects such as:
- Customer base
- Products
- Markets
- Strategy
- Risks
- etc
Management review’s key values are that it reviews your QMS ensuring
- Suitability
- Effectiveness
- Alignment to company goals
Without an effective management review process, your QMS could be outdated and not offering appropriate support.
Common issues with Management review
Of course, Management review isn’t a panacea. Without due care and attention, things can go wrong. Common issues that can be experienced include:
- Lack of buy-in
- Lack of relevance
- Failure to capture data
- Failure to follow up
- Focus on ISO and not on adding value
The key to avoiding issues is to ensure your process adds value. Once your team sees that, they’ll be keen to be involved!
Summary
Hopefully, you’ve enjoyed our article on management review. We’d love to hear feedback both on out article and on how you manage your reviews in your business. Got some tips or tricks, perhaps on how to drive engagement with your business or types of evidence you’ve used to secure your ISO:9001 certification – we’d love to hear. Please use our comments section or message us on Twitter.
